-
Study
-
Quick Links
- Open Days & Events
- Fixed Block Degrees
- Real-World Learning
- Unlock Your Potential
- Tuition Fees, Funding & Scholarships
- Still Time to Apply
-
Undergraduate
- Application Guides
- UCAS Exhibitions
- Extended Degrees
- School & College Outreach
- Parents & Guardians
-
Postgraduate
- Application Guide
- Postgraduate Research Degrees
- Flexible Learning
- Change Direction
- Register your Interest
-
Student Life
- Students' Union
- The Hub - Student Blog
- Accommodation
- Northumbria Sport
- Support for Students
-
Learning Experience
- Real-World Learning
- Research-enriched learning
- Graduate Futures
- The Business Clinic
- Study Abroad
-
-
International
International
Ideally situated in the 5th best student city in the UK (QS Best Student Cities 2026), Northumbria University is a UK Top 40 University (Complete University Guide 2026) with a diverse community of 34,500 students from over 140 countries.
Discover more-
International Students
- Information for International Students
- Northumbria and your Country
- International Student Events
- Guide to Applying
- Entry Requirements and Education Country Agents
- Global Offices and Regional Teams
- English Requirements
- English Language Centre
- International student support
- Cost of Living
-
International Fees and Funding
- International Undergraduate Fees
- International Undergraduate Funding
- International Masters Fees
- International Masters Funding
- International Postgraduate Research Fees
- International Postgraduate Research Funding
- Useful Financial Information
-
International Partners
- Agent and Representatives Network
- Global Partnerships
- Global Community
-
International Mobility
- Study Abroad
- Information for Incoming Exchange Students
-
-
Business
Business
Northumbria University is proud to offer a range of Professional, Statutory and Regulatory Body (PSRB) approved & accredited courses and programmes. Explore our list of courses and programmes under our Education and Training page.
Discover more-
Business Quick Links
- Contact Us
- Business Events
- Research and Consultancy
- Education and Training
- Workforce Development Courses
- Join our mailing list
-
-
Research
Research
Northumbria is a research-rich, business-focused, professional university with a global reputation for academic quality. We conduct ground-breaking research that is responsive to the science & technology, health & well being, economic and social and arts & cultural needs for the communities
Discover more-
Quick Links
- Research Peaks of Excellence
- Academic Departments
- Research Staff
- Postgraduate Research Studentships
- Research Events
-
Research at Northumbria
- Interdisciplinary Research Themes
- Research Impact
- REF
- Partners and Collaborators
-
Support for Researchers
- Research and Innovation Services Staff
- Researcher Development and Training
- Ethics, Integrity, and Trusted Research
- University Library
- Vice Chancellors Fellows
-
Research Degrees
- Postgraduate Research Overview
- Doctoral Training Partnerships and Centres
- Academic Departments
-
Research Culture
- Research Culture
- Research Culture Action Plan
- Concordats and Commitments
-
-
About Us
-
About Northumbria
- Our Strategy
- Our Staff
- Our Schools
- Place and Partnerships
- Leadership & Governance
- University Services
- Northumbria History
- Contact us
- Online Shop
-
-
Alumni
Alumni
Northumbria University is renowned for the calibre of its business-ready graduates. Our alumni network has over 253,000 graduates based in 178 countries worldwide in a range of sectors, our alumni are making a real impact on the world.
Discover more - Work For Us
Latest News and Features
More events
Upcoming events

IP Showcase
Northumbria University
-

Fuel & Find: A KTP Breakfast Forum
Room 304 - Business Hub, Sandyford Building
-

Supporting Survivors of Sexual Violence and Abuse Conference 2026
CCE1 - City Campus East 1
-

-
REVEAL Architecture
Architecture Studios

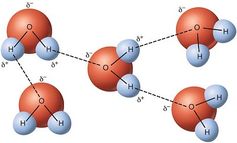 How can this be? The structure of a water molecule, H₂O, is very interesting and can be pictured like a sort of arrow tip, with the two hydrogen atoms flanking the oxygen atom at the top. The electrons in the molecule tend to be distributed in a rather asymmetric way, making the oxygen side negatively charged relative to the hydrogen side. This simple structural feature leads to a kind of interaction between water molecules known as hydrogen bonding, in which the opposite charges attract each other.
How can this be? The structure of a water molecule, H₂O, is very interesting and can be pictured like a sort of arrow tip, with the two hydrogen atoms flanking the oxygen atom at the top. The electrons in the molecule tend to be distributed in a rather asymmetric way, making the oxygen side negatively charged relative to the hydrogen side. This simple structural feature leads to a kind of interaction between water molecules known as hydrogen bonding, in which the opposite charges attract each other.






